|
4/8/2023 0 Comments Complex ion bonding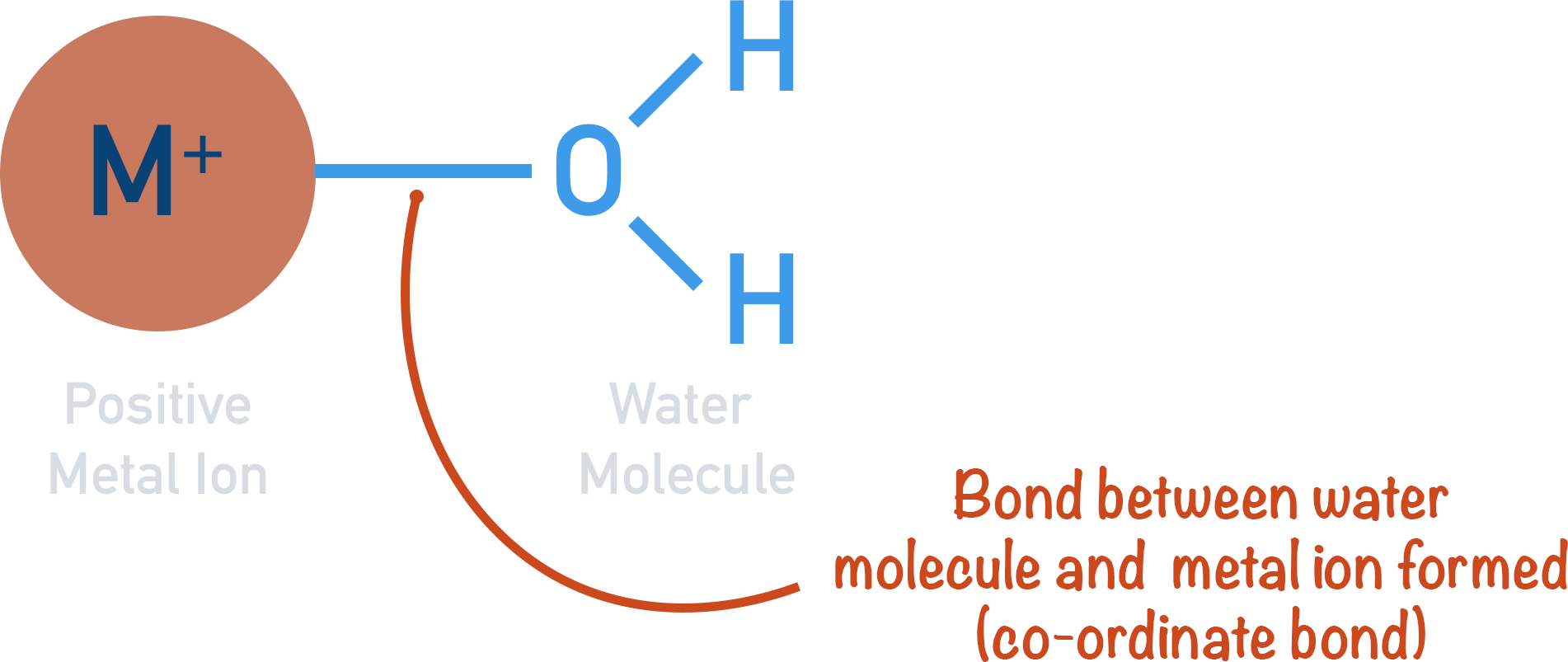
Common ligands are ammonia (NH3), water (H2O) and halide ions (Cl, Br etc). A complex ion typically consist of a tra. The ligands are neutral molecules or ions that contain lone pairs of electrons that can bond with the metal ion. This chemistry video tutorial provides a basic introduction into complex ions, ligands, and coordination compounds. (In some cases, the bonding is actually more complicated than that. Ligands are also called complexing agents. Ligands are Lewis bases - they contain at least one pair of electrons to donate to a metal atom/ion. These can be considered to be attached to the central ion by co-ordinate (dative covalent) bonds. A coordination complex is the product of a Lewis acid-base reaction in which neutral molecules or anions (called ligands) bond to a central metal atom (or ion) by coordinate covalent bonds. You probably should spend a fair amount of time discussing the σ and π interactions between manganese and the cyanido ligands including pointing out the high covalency of the $\ce$. A complex ion is composed of two things, a metal ion and compounds called ligands. A complex ion has a metal ion at its centre with a number of other molecules or ions surrounding it. It is important to understand the bonding situation in complexes which can be difficult and, yes, complex.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
